Copied
ECCO’24 Nutritional Highlights: How, When, and Why of Dietary Treatments for IBD
Learn which nutritional therapy approach is recommended depending on the IBD course and what emerging science shows on patient-friendly alternatives to enteral nutrition.

In February 2024, Nutritional Therapy for IBD attended the 19th Congress of ECCO (ECCO’24) in Stockholm, Sweden. Find out more about what the latest research says about which diets fit the disease course of IBD and considerations before implementing them in clinical practice (Part 1).
When Should Nutritional Interventions Be Used in the Course of IBD?
This year’s congress theme was ‘Crossing Borders in IBD’, aligned with the new REACH strategy launched at the congress. The strategy is an acronym of five clear goals to shape current and future ECCO activities: rapid diagnosis and treatment; equitable access to IBD care; attain sustainable IBD care; causes of IBD; and holistic IBD care.
Diet is important in IBD management because malnutrition (under and over-nutrition) leads to poor clinical and surgical outcomes in IBD. Dr Emma Halmos, a senior research dietitian at Monash University in Australia, highlighted that early nutritional and dietary assessment and intervention with a dietitian, even just education on where you can use diet and where you cannot use diet during the course of IBD, is important for improving health outcomes and quality of life, in particular in patients with IBD placed on restrictive diets.1 Halmos highlighted that nutritional therapy can fit in numerous places in children and adults with IBD care: at diagnosis, to induce remission, with uncomplicated or complicated disease, with medications to improve response, to manage non-inflammatory symptoms, and as salvage therapy.2,3
At any stage, dietitians are central in helping implement dietary therapies and improve their efficacy, correcting nutritional deficiencies (in particular vitamin D, vitamin B1, folate, zinc, iron, calcium, and magnesium),4-8 and identifying food avoidance and restrictive dietary behaviours driven by the fear of obstruction in stricturing Crohn’s disease or based on misinformation on dietary treatments for IBD.9 In the words of Halmos: “If you don’t have a dietitian, get one!”. Dietitians of ECCO are working with an upcoming consensus on diet and nutrition in IBD.
The dietary management of non-inflammatory symptoms deserves special attention at any stage of the disease2,3 Non-inflammatory symptoms include functional gut symptoms (“IBS-like” symptoms and constipation), bile acid diarrhea, pancreatic exocrine insufficiency, oxalate stones, and symptomatic strictures. “IBS-like” symptoms are related to continuing abnormality of intestinal permeability due to persistent underlying inflammation and have been reported to be 3-fold more common in patients with quiescent or inactive IBD compared to the general population.10
The fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAP)-gentle diet -consisting with the reduction of a few foods that are highly concentrated in FODMAPs- and dietary laxatives may help manage functional gut symptoms in patients with IBD.11 When it comes to dietary fiber advice, the one-size-fits-all advice of recommending low-residue diets to all patients with IBD should be replaced with a personalized diet plan. Halmos highlighted that the standard advice of avoiding dietary fiber during a flare is not recommended or supported by IBD guidelines except in the context of obstructive symptoms, including abdominal cramping, watery stools, straining, urgency, and a sensation of incomplete evacuation.

Source: Halmos’ presentation at ECCO’24.
Nutrition Assessment and Treatment in Patients with IBD Undergoing Surgery: What Is New?
According to Julie Vanderstappen, clinical IBD dietitian from University Hospitals Leuven in Belgium, diet has a role in IBD prevention, therapy in active disease, screening for malnutrition and nutritional deficiencies and treating IBD complications. However, some gaps exist relating to nutrition in IBD as 41% of the gastroenterologists rated their knowledge of nutrition in IBD as “very good” compared with 87% of dietitians and 16% of nurses.12 While psychosocial factors relating to food are important for people with IBD, limited research considers them. One validated tool that can help measure the psychosocial aspects of eating and drinking in IBD is the FRQoL-29 short and self-report questionnaire.13
Regarding diet throughout surgery, Vanderstappen acknowledged that it is important to perform an assessment of the perioperative nutritional status of patients with IBD and exclusive enteral nutrition (EEN), partial enteral nutrition (PEN), and parenteral nutrition (PN) may help improve nutritional status in patients with CD. Halmos every patient planned for surgery needs nutritional therapy in the form of at least two oral nutritional supplements for seven days prior to the surgery.
Nutritional status should be assessed before and after surgery, and personalized nutritional support provided in a timely manner may enhance recovery of the patient.14,15 Clinical assessment, assessment of dietary intake, anthropometric measurements, biomarkers of nutritional status, and validated screening tools are useful tools for preoperative nutritional assessment.

Source: Vanderstappen’s presentation at ECCO’24.
Iris Dotan and Lihi Godny from the Rabin Medical Center (Israel) updated dietetic management of the ileal pouch. Diet and microbiome might play a role in pouchitis based on the rationale that reduced microbial diversity precedes pouchitis, and fruit consumption may protect against pouchitis by shaping gut microbiota composition.16,17
The presence of a more balanced gut microbiota composition in patients with normal pouch has prompted Dotan and Godny to explore the Mediterranean Diet as an option for patients after pouch surgery. One mechanism by which the MED diet may help patients after surgery is through shaping the gut microbiota towards a protective profile (e.g., rich in butyrate producers Faecalibacterium and Lachnospira). A microbiota-targeted MED-based intervention might be used in patients after surgery as a means to prevent new episodes of pouchitis, as an add-on to antibiotics, as a potential alternative to replace rounds of antibiotics, or as a strategy to improve metabolic health.18 Other dietary therapies for patients after surgery are on the way. While foods rich in fermentable fibers and micronutrients might play a beneficial role in pouchitis,19 the impact of elemental diet therapy for reducing symptoms and inducing endoscopic remission in patients with active pouchitis remains inconclusive.20,21
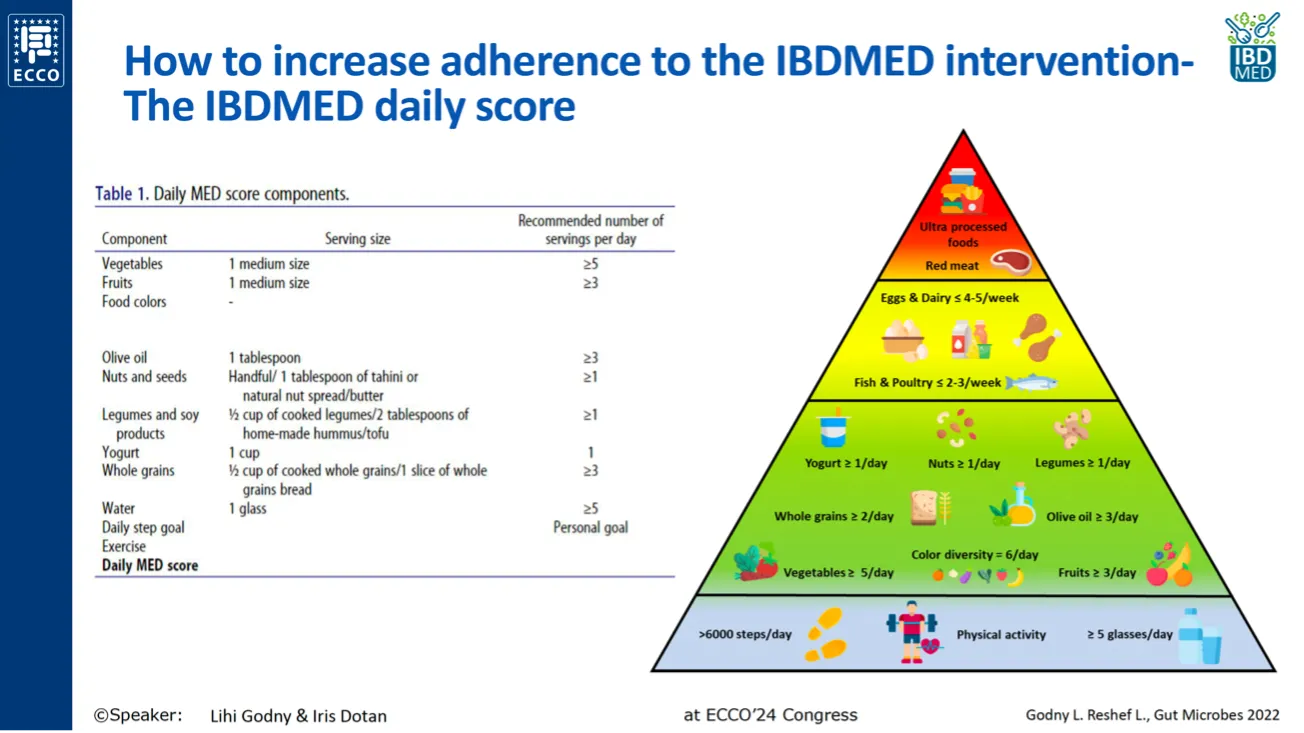
Mediterranean diet components with a protective role in IBD.
Source: Dotan and Godny’s presentation at ECCO’24. Based on the reference 22.
Exclusive Enteral Nutrition: From Theory to Practice
Diet is integrated into the updated ECCO CD guidelines, which will focus on early effective treatment, advocate for personalized medical therapy, incorporate new drugs, downgrade some old treatments, and substitute “conventional” with “evidence-based and effective” when referring to treatments. The new CD ECCO guideline states:
- EEN can be used as induction therapy in children and adults with mild-to-moderate CD who are motivated to adhere to dietary therapy, have access to dietetic support, and prefer to avoid corticosteroids
- All patients with CD should have access to dietary services, especially during disease flare
- Dietary intervention should be considered based on disease activity, the patient’s motivation, the current evidence, and the availability of dietetic support
- PEN might be considered as a strategy for maintaining remission, with or without additional medication, in a subset of patients who are willing and able to tolerate the formula with routine monitoring
Dr Richard Hansen, a medical doctor from the University of Dundee (United Kingdom) and researcher into the role of the gastrointestinal mucosal microbiota in paediatric IBD, focused on what is changing in diet and paediatric CD. The benefits of EEN for CD are not new and date back from 197323 and Hansen highlighted that EEN shows a paradoxical mechanism of action that is distinct from all drugs: while it favours an altered gut microbiota due to its lack of fiber, it improves inflammation. In children and adolescents with moderate-severe CD who have not finished their growth, EEN is the induction therapy of first choice for 6-8 weeks.24 EEN shows an overall remission rate of 52% and a completion rate higher if the formula is administered orally (74%) versus nasogastric tube (28%).25
However, one of the limitations of EEN is its suboptimal response in a subset of children with CD, which is explained in part by poor compliance that threatens the traditional role of EEN as induction therapy.26,27 Emerging alternatives to obtain the benefits of EEN without the tolerance issues include: 1) reducing to PEN alongside some food (known as Crohn’s Disease Exclusion Diet) and 2) Replace all EEN with food, which is known as the CD treatment-with-eating diet (CD-TREAT).

Hansen also presented relevant data on using EEN in combination with biologics (e.g., infliximab), which appears to be more effective at inducing and maintaining remission among patients with CD than infliximab monotherapy.28 Also, some evidence suggests pre-surgical EEN may improve the length of post-surgical hospitalization and the risk of post-surgical infectious complications in patients with CD.29
How Can Patients with IBD Obtain the Benefit of EEN Without Tolerance Issues? CD Exclusion Diet and Other Patient-Friendly Diets
During ECCO'24, Nestlé Health Science organized a symposium on Nutritional Management of Crohn’s disease. The session focused on how and why nutritional therapy should be incorporated in the course of Crohn's disease, the role of enteral nutrition for induction when combined with exclusion diets, for complicated disease, as a maintenance therapy after induction, to prevent and optimize postoperative outcomes, and combined with biologics, and new studies supporting the efficacy of patient-friendly diets, including CDED.
Rotem Sigall-Boneh, a clinical and research dietitian from Israel, highlighted that the Crohn’s Disease Exclusion Diet (CDED) is the most extensively researched diet, showing high tolerability and efficacy for inducing clinical and biochemical remission and maintenance of remission in children and adults with mild-to-moderate Crohn’s disease as supported by recent systematic reviews30 and clinical guidelines.31 CDED consists of three phases and dietitians are central in educating patients and improving their adherence to the diet in order to achieve better health outcomes.26

Limitations of the CDED are that its long-term effectiveness in IBD remission is limited, and it is not known its role in severe and complicated CD together with other disease phenotypes that might benefit from using it and its efficacy when combined with biologics.26 Sigall-Boneh shared new data showing that 2 weeks of EEN followed by CDED and PEN for 24 weeks successfully maintained remission for up to 52 weeks in children with mild-to-severe CD and was accompanied with an improved nutritional status. It remains to be seen how CDED monotherapy works in long-term remission for some patients.

Click image to open PDF
Sigall-Boneh summarized available patient-friendly novel dietary interventions for IBD with variable efficacy including the specific carbohydrate diet, the Mediterranean diet, the Crohn’s disease-TREAT diet, the IBD-AID diet and autoimmune diet. Ongoing clinical trials are studying the effects of diet on intestinal microbiota changes and inflammation in healthy individuals and patients with UC (the Food influence on the Intestinal microbiota or FIT trial) and identifying the role of a low-emulsifier diet in CD (the ADDapt trial).

Source: Sigall-Boneh’s presentation at ECCO’24.
Patients with CD in clinical and biochemical remission tend to be less adherent to the beneficial components of the Mediterranean diet. It is also worth noting that adherence to the Mediterranean diet has been associated with lower faecal calprotectin levels at baseline and at 3 months of follow-up.32
Variants of the Mediterranean diet might also offer benefits for patients with IBD. After following almost 190,000 participants from the United Kingdom for 11 years, S. Ye and colleagues from the Third Xiangya Hospital of Central South University in China suggested that the Mediterranean-DASH Diet Intervention for Neurodegenerative Delay, or MIND diet, which combines foods from the Mediterranean and DASH diets that had been shown to benefit brain health, may help to prevent IBD. In particular, a high intake of olive oil was associated with a low risk of Crohn’s disease.33
Some dietary patterns can shift the gut microbiome towards a composition that worsens the outcomes of patients with IBD. This is the case with the sulfur microbial diet, high in processed meats and low in vegetables and legumes, which enriches sulfur-metabolizing microbes in the gut. This results in an increase in the microbial production of hydrogen sulfide (H2S) that is associated with an elevated risk of bowel resection surgery and colorectal cancer in patients with IBD (up to 76% and 268% higher risk, respectively), according to poster findings from Jie Chen and colleagues from the Third Xiangya Hospital of Central South University in China.34
According to the Roediger pathogenic hypothesis for UC, the prolonged production of nitric oxide (NO) and hydrogen sulfide (H2S) from protein fermentation by colonic bacteria may alter gut microbiota composition and function and inhibit butyrate metabolism within the colonocyte, ultimately resulting in a breakdown of epithelial barrier, which is central to the development of UC.35,36

Source: Halmos’ presentation at ECCO’24.
Why Adherence to Dietary Therapy Matters
The extent to which a patient with IBD follows nutritional therapy corresponding with agreed recommendations from a healthcare provider (adherence or patient’s involvement in diet therapy) is central to the treatment effect of diet. Dr Alice Day, an Advanced Research Dietitian undertaking research in diet therapies for IBD, highlighted that evaluating adherence should run parallel to evaluating intent and efficacy and examination of mechanisms of action of diet within the intraluminal environment. Methodological improvement is needed in future IBD diet trials as 2 out of 3 existing prospective IBD diet trials have reported on dietary adherence, and less than 1 out of 3 studies involved a dietitian to design and educate on diet strategy.
Dietitians must support and monitor adherence as part of their usual clinical practice through IBD diet assessment, nutritional adequacy, dietary perceptions and beliefs, and diet tolerability. Interestingly, how a dietitian delivers advice to the patient can impact the success of dietary therapy. Laura Portmann and colleagues from the Queen Elizabeth Hospital and Basil Hetzel Institute in Australia showed that dietitian-led comprehensive written and verbal dietary education is better than generalized written advice for achieving dietary changes in adults with UC.

Click Image to open PDF
When it comes to adherence to CDED diet, Fleur Wijers from Amsterdam University Medical Centers and colleagues reported findings from a telephone interview showing that CDED is feasible to follow for patients (paediatric and adult) with CD as part of their medical treatment, being better assessed by adults than for children and their parents. One of the aspects that patients with CD valued the most is the availability of tools that allow them to follow the diet. For instance, the ModuLife patient platform allows to support patients through each phase of the CDED.

Click image to open PDF
Continue reading in the second part of ECCO highlights
Further reading:
- Schreiner P, Yilmaz B, Rossel JB, et al. Vegetarian or gluten-free diets in patients with inflammatory bowel disease are associated with lower psychological well-being and a different gut microbiota, but no beneficial effects on the course of the disease. United European Gastroenterol J. 2019; 7(6):767-781. doi: 10.1177/2050640619841249.
- FitzpatrickJ A, Melton SL, Yao CK, et al. Dietary management of adults with IBD – the emerging role of dietary therapy. Nat Rev Gastroenterol Hepatol. 2022;19(10):652-669.doi: 10.1038/s41575-022-00619-5.
- Halmos EP, Godny L, Vanderstappen J, et al. Role of diet in prevention versus treatment of Crohn’s disease and ulcerative colitis. Front Gastroenterol. Published Online First: 09 January 2024. doi: 10.1136/flgastro-2023-102417
- Lytvyak E, Sutton RT, Dieleman LA, et al. Management of inflammatory bowel disease patients with clinical care pathways reduces emergency department utilization. Crohns Colitis 360. 2020; 2(4):otaa080. doi: 10.1093/crocol/otaa080.
- Dignass AU, Gasche C, Bettenworth D, et al. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. J Crohns Colitis. 2015; 9(3):211-22. doi: 10.1093/ecco-jcc/jju009.
- Kilby K, Mathias H, Boisvenue L, et al. Micronutrient absorption and related outcomes in people with inflammatory bowel disease. Nutrients. 2019; 11(6):1388. doi: 10.3390/nu11061388.
- Fletcher J, Cooper SC, Ghosh S, et al. The role of vitamin D in inflammatory bowel disease: mechanism to management. Nutrients. 2019; 11(5):1019. doi: 10.3390/nu11051019.
- Vaghari-Tabari M, Jafari-Gharabaghlou D, et al. Zinc and selenium in inflammatory bowel disease: trace elements with key roles? Biol Trace Elem Res. 2021; 199(9):3190-3204. doi: 10.1007/s12011-020-02444-w.
- Day AS, Yao CK, Costello SP, et al. Food avoidance, restrictive eating behaviour and association with quality of life in adults with inflammatory bowel disease: A systematic scoping review. Appetite. 2021; 167:105650. doi: 10.1016/j.appet.2021.105650.
- Farrokhyar F, Marshall JK, Easterbrook B, et al. Functional gastrointestinal disorders and mood disorders in patients with inactive inflammatory bowel disease: prevalence and impact on health. Inflamm Bowel Dis. 2006; 12(1):38-46. doi: 10.1097/01.mib.0000195391.49762.89.
- Halmos EP, Gibson PR. Controversies and reality of the FODMAP diet for patients with irritable bowel syndrome. J Gastroenterol Hepatol. 2019; 34(7):1134-1142. doi: 10.1111/jgh.14650.
- Tinsley A, Ehrlich OG, Hwang C, et al. Knowledge, attitudes, and beliefs regarding the role of nutrition in IBD among patients and providers. Inflamm Bowrl Dis. 2016; 22(10):2474-81. doi: 10.1097/MIB.0000000000000901.
- Hughes LD, King L, Morgan M, et al. Food-related quality of life in inflammatory bowel disease: development and validation of a questionnaire. J Crohns Colitis. 2016; 10(2):194-201. doi: 10.1093/ecco-jcc/jjv192.
- Adamina M, Bonovas S, Raine T, et al. ECCO guidelines on therapeutics in Crohn’s disease: surgical treatment. J Crohns Colitis. 2020; 14(2):155-168. doi: 10.1093/ecco-jcc/jjz187.
- Weimann A, Braga M, Carli F, et al. ESPEN practical guideline: clinical nutrition in surgery. Clin Nutr. 2021; 40(7):4745-4761. doi: 10.1016/j.clnu.2021.03.031.
- Dubinsky V, Reshef L, Rabinowitz K, et al. Dysbiosis in metabolic genes of the gut microbiomes of patients with an ileo-anal pouch resembles that observed in Crohn’s disease. mSystems. 2021; 6(2):e00984-20. doi: 10.1128/mSystems.00984-20.
- Godny L, Maharshak N, Reshef L, et al. Fruit consumption is associated with alterations in microbial composition and lower rates of pouchitis. J Crohns Colitis. 2019; 13(10):1265-1272. doi: 10.1093/ecco-jcc/jjz053.
- Godny L, Dotan I. Is the Mediterranean Diet in inflammatory bowel diseases ready for prime time? J Can Assoc Gastroenterol. 2023; 7(1):97-103. doi: 10.1093/jcag.gwad041.
- Ardalan ZS, Yao CK, Sparrow MP, et al. Review article: the impact of diet on ileoanal pouch function and on the pathogenesis of pouchitis. Aliment Pharmacol Ther. 2020; 52(8):1323-1340. doi: 10.1111/apt.16085.
- McLaughlin SD, Culkin A, Cole J, et al. Exclusive elemental diet impacts on the gastrointestinal microbiota and improves symptoms in patients with chronic pouchitis. J Crohns Colitis. 2013; 7(6):460-6. doi: 10.1016/j.crohns.2012.07.009.
- Yamamoto T. Elemental diet therapy for pouchitis following restorative proctocolectomy for ulcerative colitis. J Crohns Colitis. 2013; 7(4):e155. doi: 10.1016/j.crohns.2012.09.004.
- Godny L, Reshef L, Sharar Fischler T, et al. Increasing adherence to the Mediterranean diet and lifestyle is associated with reduced fecal calprotectin and intra-individual changes in microbial composition of healthy subjects. Gut Microbes. 2022; 14(1):2120749. doi: 10.1080/19490976.2022.2120749.
- Giorgini GL, Stephens RV, Thayer Jr WR. The use of “medical by-pass” in the therapy of Crohn’s disease: report of a case. Am J Dig Dis. 1973; 18(2):153-7. doi: 10.1007/BF01073162.
- Ruemmele FM, Veres G, Kolho KL, et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J Crohns Colitis. 2014; 8(10):1179-207. doi: 10.1016/j.crohns.2014.04.005.
- Wands DIF, Gianolio L, Wilson DC, et al. Nationwide real-world exclusive enteral nutrition practice over time: persistence of use as induction for pediatric Crohn’s disease and emerging combination strategy with biologics. Inflamm Bowel Dis. 2023; izad167. doi: 10.1093/ibd/izad167.
- Sigall Boneh R, Westoby C, Oseran I, et al. The Crohn’s Disease Exclusion Diet: a comprehensive review of evidence, implementation strategies, practical guidance, and future directions. Inflamm Bowel Dis. 2023. doi: 10.1093/ibd/izad255.
- Mckirdy S, Russell RK, Svolos V, et al. The impact of compliance during exclusive enteral nutrition on faecal calprotectin in children with Crohn disease. J Pediatr Gastroenterol Nutr. 2022; 74(6):801-804. doi: 10.1097/MPG.0000000000003425.
- Nguyen DL, Palmer LB, Nguyen ET, et al. Specialized enteral nutrition therapy in Crohn’s disease patients on maintenance infliximab therapy: a meta-analysis. Therap Adv Gastroenterol. 2015; 8(4):168-75. doi: 10.1177/1756283X15578607.
- Lomer MCE, Wilson B, Wall CL. British Dietetic Association consensus guidelines on the nutritional assessment and dietary management of patients with inflammatory bowel disease. J Hum Nutr Diet. 2023; 36(1):336-377. doi: 10.1111/jhn.13054.
- Zhu Z, Lei Y, Lin Z, et al. Effects of Crohn’s disease exclusion diet on remission: a systematic review. Therap Adv Gastroenterol. 2023; 16:17562848231184056. doi: 10.1177/17562848231184056.
- Bischoff SC, Bager P, Escher J, et al. ESPEN guideline on clinical nutrition in inflammatory bowel disease. Clin Nutr. 2023; 42(3):352-379. doi: 10.1016/j.clnu.2022.12.004.
- L Mor-Sasson, C Sarbagili-Shabat, S Sherf-Dagan, et al. Adherence to a Mediterranean dietary pattern in patients with Crohn’s disease in remission is associated with lower fecal calprotectin levels. Journal of Crohn’s and Colitis. 2024; 18(Supplement_1):i1981-i1982. https://doi.org/10.1093/ecco-jcc/jjad212.1232.
- S Ye, Y Sun, X Chen, et al. Mediterranean dietary approaches to stop hypertension intervention for neurodegenerative delay (MIND) diet can reduce Inflammatory Bowel Disease risk: a prospective cohort study. Journal of Crohn’s and Colitis. 2024; 18(Supplement_1):i2064. https://doi.org/10.1093/ecco-jcc/jjad212.1284.
- J Chen, L Dan, Y Sun, et al. The sulfur microbial diet and prognosis of individuals with Inflammatory Bowel Disease in a prospective cohort study. Journal of Crohn’s and Colitis. 2024; 18(Supplement_1):i2059. https://doi.org/10.1093/ecco-jcc/jjad212.1280.
- Roediger WEW. Review article: nitric oxide from dysbiotic bacterial respiration of nitrate in the pathogenesis and as a target for therapy of ulcerative colitis. Aliment Pharmacol Ther. 2008; 27(7):531-41. doi: 10.1111/j.1365-2036.2008.03612.x.
- Pitcher MC, Beatty ER, Cummings JH. The contribution of sulphate reducing bacteria and 5-aminosalicylic acid to faecal sulphide in patients with ulcerative colitis. Gut. 2000; 46(1):64-72
Stay informed of the latest news in nutrition and IBD




Support our Mission
Your donation will help us to enhance the well-being and health outcomes of patients with IBD.
Donate Donate
Donate
